Solvent:
- Solvent is a substance which dissolves the solute in the solution.
- It is used to dissolve the solute.
- Water is used to dissolve much number of substances so water is known as the universal solvent.
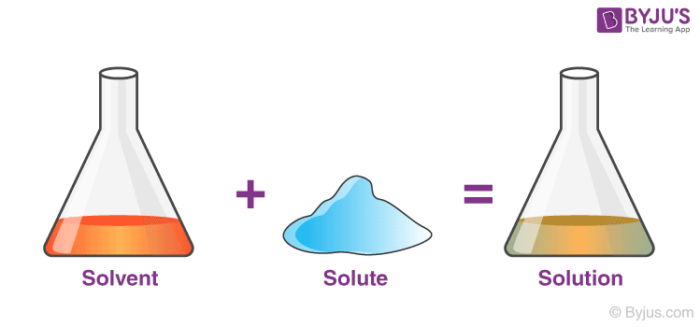
Solute + Solvent = Solution
For example, The sugar solution can be made by dissolving sugar crystals in water. Here, Sugar crystals are solute, water is the solvent used.
Related Posts
Classification:
The solvent can be classified as
- Polar solvent
- Non Polar solvent
“Like dissolves Like”
- Polar solvent:
The substance which dissolves in polar solvent is known as polar molecules.
For Example, Water.
2. Non Polar solvent:
The substance which do not dissolve in water is known as non polar molecules.
For Example, The aromatic compounds such as alcohols, amines, ketones, esters etc.,
Physicochemical properties of solvent:
Evaporation rate : The time required to change of solvent from liquid to vapour.
Vapour pressure : The pressure of a liquid is in equilibrium with vapor at a given temperature.
Boiling point : The temperature at which the vapor pressure equal to the atmospheric pressure.
Explosive limit : The potential for explosion.
Health hazards of solvents:
- The short term exposure to solvents may cause irritation in eye, lungs and skin, headache, dizziness.
- Organic solvents are carcinogenic, reproductive hazards and neurotoxins.
- The long term exposure to solvents may cause liver, brain and kidney damage.
Application of solvents:
- Solvents are used to dissolve paints, pigments.
- It is used as thinning agent.
- It is used to clean automobile parts, tools and electronic goods.