Photochemistry Definition:
Photochemistry deals with the study of chemical reaction by the absorption of light radiation. It should have the sufficient energy to raise an electron from the ground state to an excited state.
Consequences of Light Absorption
According to the Grotthus- Draper law, the light absorbed by a system can cause chemical reaction but not all the light absorbed must cause chemical reactions.
Firstly, the light absorbed by the system causes decrease in the intensity of incident radiation. This event is governed by Beer – Lambert law.
Related Posts
Secondly, the light absorbed may be re emitted immediately in or two two steps. This phenomenon is known as fluorescence. Sometimes the light absorbed may be given out slowly or even at the end of the process. This is known as phosphorescence. The phenomenon of phosphorescence and fluorescence was explained by Jablonski diagram.
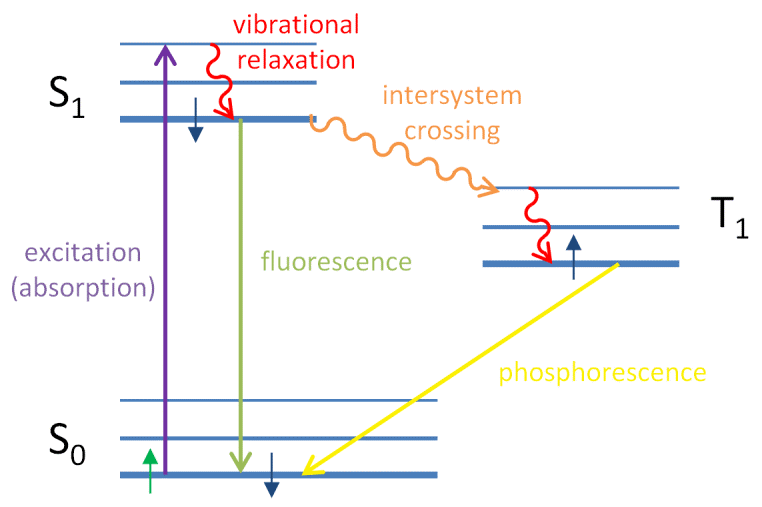
quantum Yield
The quantum yield of a photochemical process is defined as
φ = Number of molecules react / Number of quanta of radiation absorbed.
Photosensitization
Some reactions may occur does not by the absorption of light by one of the reactants but by a third substance which transfers the absorbed energy to the reactants. This third substance itself does not undergo any change is called photosensitizer and the process is known as photosensitization.
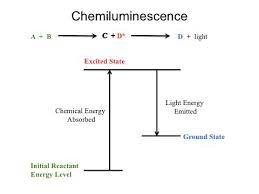
Chemiluminescence
Chemiluminescence is defined as the production of light by a chemical reaction and is thus the reverse of a photochemical reaction.