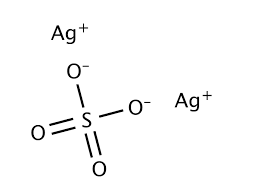
What is Silver Sulphate ?
Silver sulphate or also called silver sulphate. Its molecular formula is Ag2 SO4. This silver sulphate is fake and is more likely to change color if exposed to air. silver sulphate does not dissolve much in water and is slightly soluble. They are coated with silver to prevent them from dissolving in materials such as silver. Silver sulphate contains +2 cation silver ions and requires -2 anion silver ion to balance it.

Ads
Formula Ag2SO4
Related Posts
Lead Nitrate Definitions, Formula, Reaction ,Uses, Disadvantages and Video
What is Aliphatic Compounds , Extraction of Aliphatic Hydrocarbons , Classification , Uses of aliphatic compounds ?
What is Raman Spectroscopy, Introduction, Principle, Types of scattering, Applications ?
Electrochemistry Introduction, Principle, Conductivity, Factors affecting electrolytic conductance,Debye – Huckel and Onsager equation, Kohlraush’s law, Nernst Equation etc..?
Molar Mass 311.799 g/mol
Molecular weight 313.8
Melting Point 652 °C, 925 K, 1206 °F
Boiling Point 1085 °C, 1358 K, 1985 °F
Silver Sulphate Formula

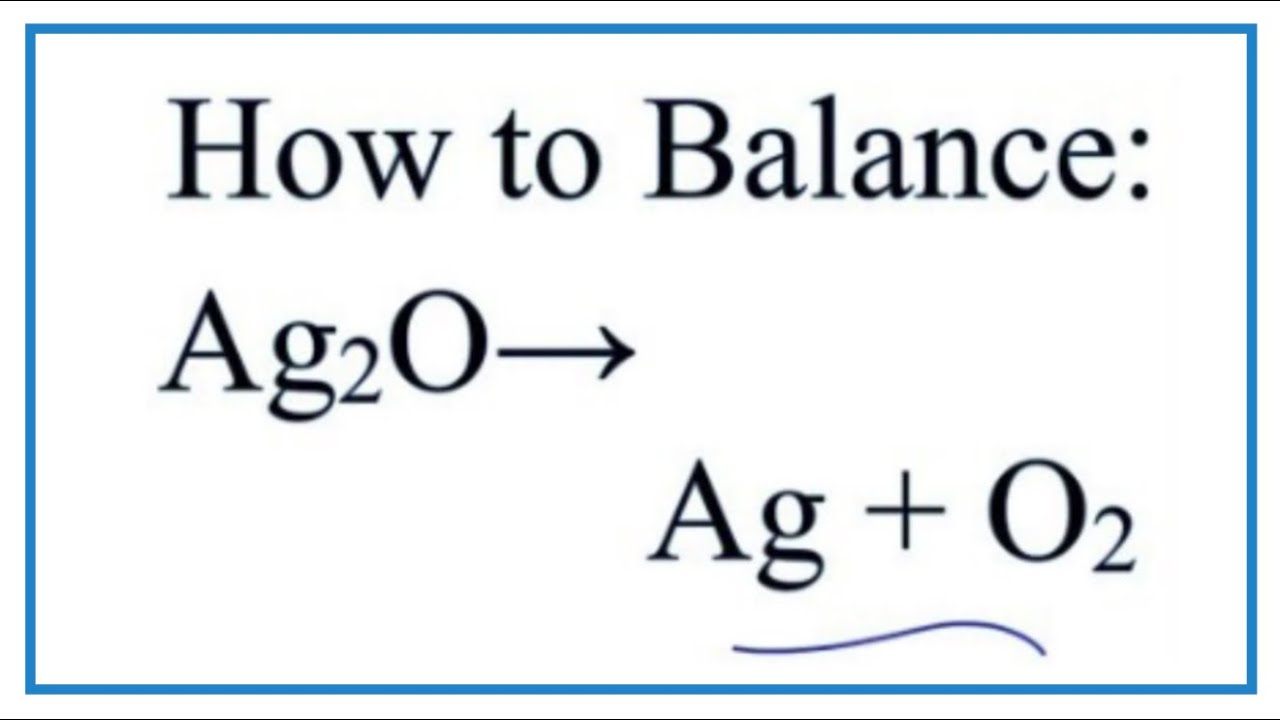
Silver Sulphate Balancing Equation
Reacting with silver sulphate gives us silver sulphate. Reacts with two ions, cation and anion.
2Ag + SO4 → Ag2SO4
Example
Silver sulphate uses :
- Silver sulfate is used to prevent other types of infections in many patients with burns.
- Silver sulphate is widely used for medical purposes.
- It has the ability to destroy bacterial cells.