What Is Alkanes :
In organic chemistry, an alkane is an acyclic saturated hydrocarbon. In other words, alkane contains carbon and hydrogen which is attached by single bond. It is having the general formula CnH2n+2. In the case of alkane, the C-atom is sp3 hybridised with four sigma bonds.
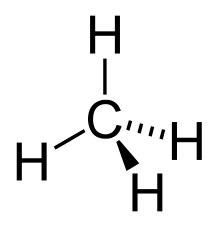
Types Of Alkanes:
1. Linear alkanes
Ads
Straight chain alkanes are indicated by the prefix ‘n-‘.
Related Posts
Lead Nitrate Definitions, Formula, Reaction ,Uses, Disadvantages and Video
What is solvent, classification, Physicochemical properties of solvent,Health hazards of solvents,Application of solvents?
Copper sulphate Formula, Structure, Uses and Video
What is inorganic chemistry , Application and examples , video :
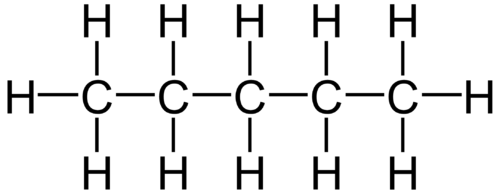
Example : n-Pentane
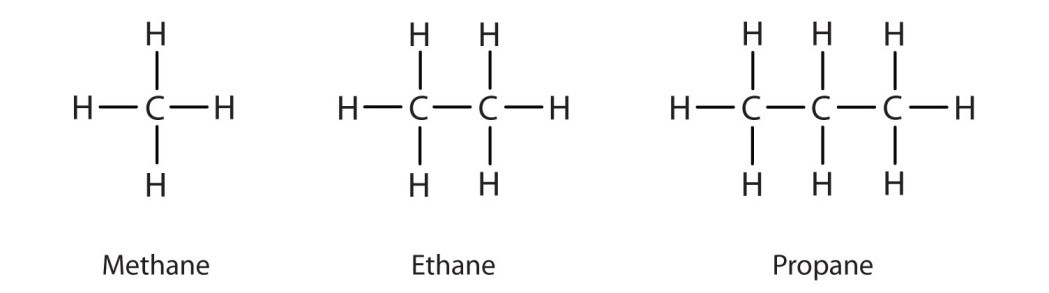
Methane – CH4
Ethane – C2H6
Propane – C3H8
Butane – C4H10
Pentane – C5H12
Hexane – C6H14
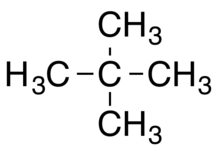
2. Branched alkane
The branched alkane is indicated by some specific prefix to distinguish from linear alkane.
For example: n-Pentane by isopentane or neopentane.
Uses of Alkanes :
- Methane and ethane are the main components of natural gas, they stored under pressure.
- The first four alkanes are used for heating and cooking purposes.
- They are used to generate electricity.
- Propane and butane gases are stored under low pressure and is known as Liquified Petroleum Gas (LPG) and is used for cooking.
- Some synthetic polymers called polyethylene and polypropylene contains number of repeated units of carbon atom.
- It’s a major component of lubricating oil and is called anti corrosive agent.
Disadvantages of Alkanes :
- It is the main contributor of Green house Gas and causes global warming.
- In humans, the high concentration of alkanes causes anaesthetic effects or narcosis.
- Highly explosive.